In the Cell Morphometrics & Mechanisms group, we develop next-generation MRI methods to uncover how cells are built, how they function, and how they change in health and disease. Our work goes beyond static images—toward dynamic, quantitative descriptions of cellular architecture, composition, and physiology.
At the core is the idea that MRI can probe the microscopic world of cells through motion. By combining advanced diffusion and flow-sensitive measurements, we capture how water and metabolites move within complex cellular environments. These motions are not random—they are shaped by membranes, organelles, macromolecules, and barriers—turning MRI into a powerful, non-invasive window on cellular structure and function.
Leveraging the enhanced spectral and spatial resolution of ultra-high field MRI, we push sensitivity to new limits, enabling us to disentangle subtle contributions from tissue chemistry, compartmental exchange, and microvascular flow. While diffusion MRI is traditionally framed as a tool for mapping geometry—cell size, shape, and orientation—we treat this as only part of the story.
Cellular systems are governed not just by structure, but by interactions: chemical composition, permeability, fluid dynamics, and the integrity of biological barriers. These factors leave measurable fingerprints in MR signals. By rethinking experimental design and modeling, we aim to decode these signals to reveal deeper physiological insights—linking microstructure to metabolism, transport, and mechanical properties.
Our vision is to transform MRI from a descriptive imaging modality into a quantitative tool for cellular biophysics in vivo, bridging scales from molecules to tissue and enabling new biomarkers of disease and treatment response.
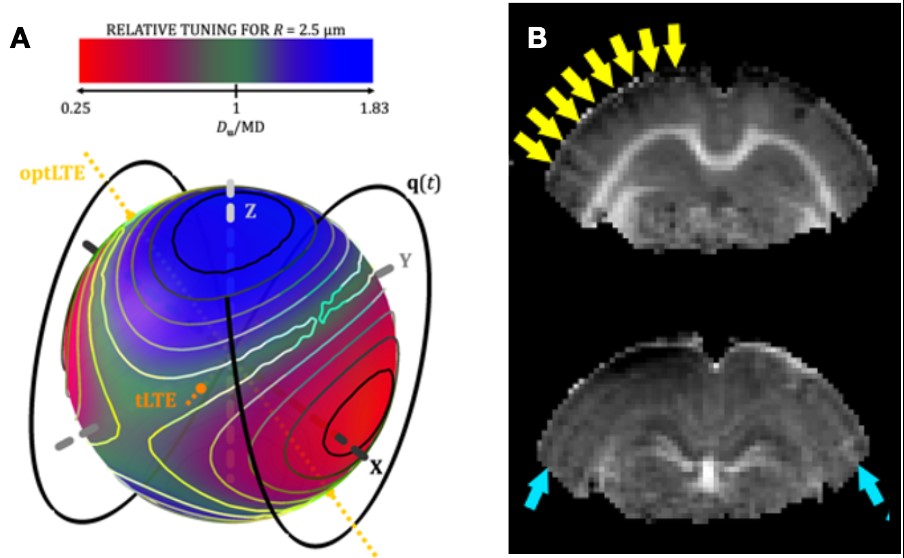
Research projects:
C-MORPH: develops advanced MRI methods to map cell-type specific properties in the living brain and spinal cord. By combining diffusion encoding, spectroscopy, and novel imaging, we disentangle microstructural and pathological processes that are otherwise entangled in conventional scans. The goal is to deliver sensitive, clinically applicable markers of disease progression, inflammation, and neurodegeneration for personalized medicine.
Funding:
Scleroseforeningen
European Research Council
Det Fri Forskningsråd
Danske Regioner





